|
This lab can be used to introduce ionic, covalent and metallic bonds and their properties. In the lab You Light Up My Life, students participate in a guided inquiry investigation which allows them to test different physical properties of given samples.Teacher guide and student activity sheet.Ĭovalent, Ionic & Metallic Bonding and Properties This simulation is unlocked and can be used by Students also becomeįamiliar with the molecular formula and geometric shape, as well as the naming Sharing of electrons when forming a covalent compound. 
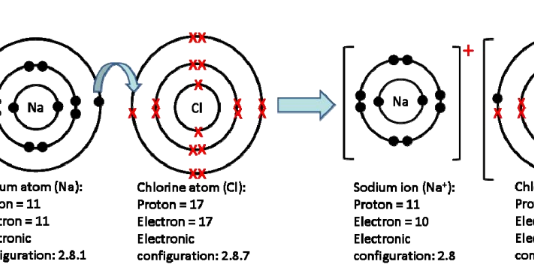
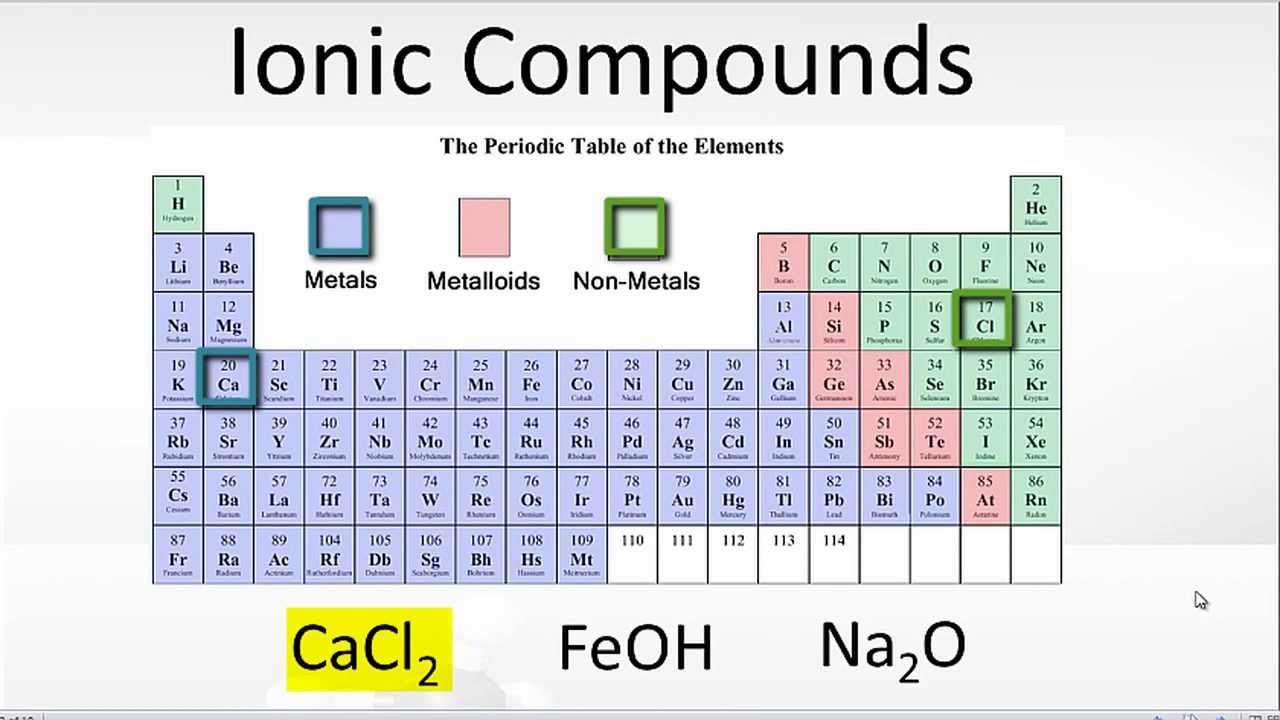
The simulation visually differentiatesīetween the transferring of electrons when forming an ionic compound and the Students interact with differentĬombinations of atoms and are tasked with determining the type of bond and the To investigate ionic and covalent bonding. Ionic & Covalent Bonding Simulation from the September Examples of ionic, covalent, and polar covalent bonds are animated, and students are given a set of compounds to predict the bonding types. Help students visualize how different chemical bonds form by using the Bonding Animation to introduce the concept of bonding.Please note that most of these resources are AACT member benefits.The teacher notes, student handouts, and additional materials can be accessed on the page for each individual activity.The number of activities you use will depend upon the level of students you are teaching.The activities shown below are listed in the order that they should be completed.Refer to the safety instructions given with each individual activity.Refer to the materials list given with each individual activity.Lesson: 8-12 class periods, depending on class level. Teacher Preparation: See individual resources. This unit supports students’ understanding of Recognize that physical properties are related to intermolecular forces.Identify the intermolecular forces present in chemical substances.Manipulate models to demonstrate molecular orientations giving rise to London dispersion forces, dipole-dipole forces and hydrogen bonds.Rank molecules in order of increasing strength of van der Waals forces, given a set of structural formulas for several compounds.Describe the unique behaviors of water molecules, and why they are important.Explain the meaning of the following: cohesion, adhesion, surface tension, and capillary action.Relate the shape of a molecule and the relative electronegativity values of its constituent atoms to the polarity of the molecule.Make the correlation between geometry, nonbonding pairs and molecular shape.Understand that the molecular shape names are descriptions of the actual shape.Describe the implications of electron pair repulsions on molecular shape.Conceptualize the impact of one electron pair domain acting upon another, and understand how those interactions result in the molecular geometries predicted by VSEPR theory.Identify that different metals have different properties. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
